
CASE Ultra is software program used in computational toxicology that identifies structural alerts related to toxicity using (Q)SAR.
CASE Ultra (Q)SAR models are validated using OECD guidelines and come with QMRF reports. Models are available for the following toxicological endpoints:
Bacterial Mutagenicity/ICH M7 more
Genotoxicity
Carcinogenicity
Skin Sensitization
Acute Toxicity
Endocrine Disruption
Reproductive Toxicity
Developmental Toxicity
Cardiotoxicity
Hepatotoxicity
Renal Toxicity
ADME
Ecotoxicity
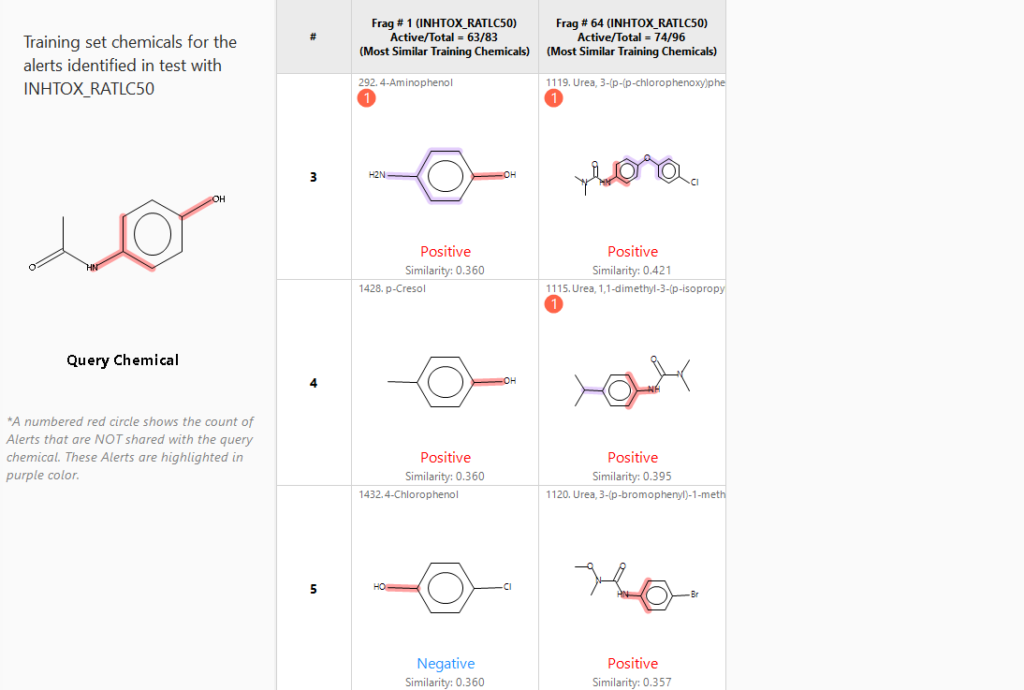
Similarity measures are automatically employed to find analogs that have the same structural alerts as the query chemical. The program also alerts the user to alerts contained by analogs that are not shared with the query chemical when appropriate.
This type of search is applied automatically by the software. When a query chemical is submitted for assessment, the connected databases are searched to determine if any experimental activity data is available. If found, the search result is reported with details such as assay conditions, dosage, duration of treatment, and species/strain information.
Similarity-based searches can be carried out for read-across purposes and to search for structural analogs to aid in expert review.
Substructure searches are sometimes used to determine the toxicity potential of specific parts of a query chemical structure. Retrieved hits with specific structural alerts can be used to determine the alerts’ relevance in the context of the query chemical. Special alert environment similarity measures are employed for finding relevant analogs that have a similar structural environment around an alert.
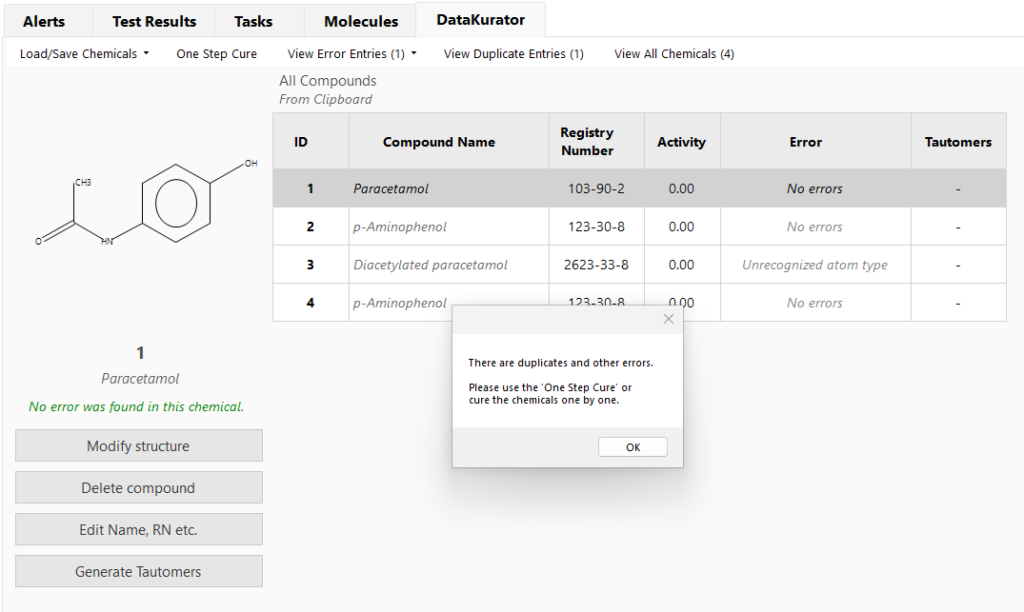
Curation of chemical data is a crucial starting step in any workflow and is carried out using the DataKurator tool. Appropriate curation is critical in getting accurate results and needed for harmonizing the structures of the query chemicals with the databases to reduce out of domain outcomes and to increase the success of the database searches. DataKurator can be used to perform batch curation of tens of thousands of chemicals at a time.
Software packages are customized to meet the needs of the user. Factors that affect pricing include length of license, number of endpoints, and number of users. Please contact us to request a quote.